
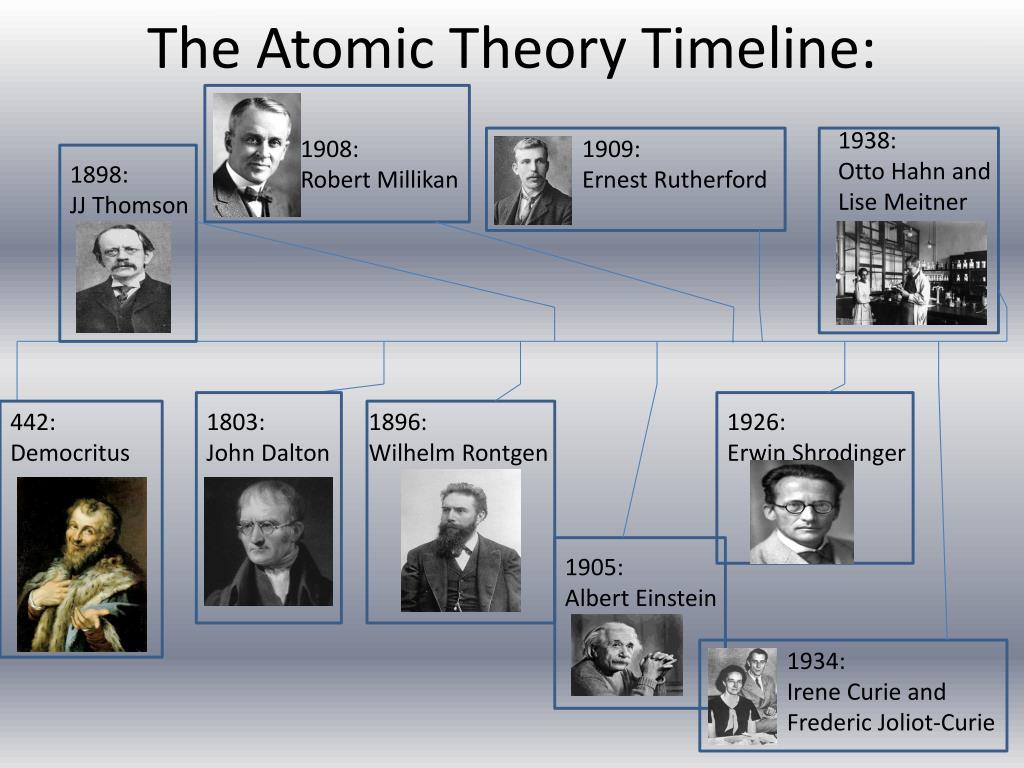
The lesson covers the concept of models, a brief overview of atoms, a printable of the atomic theory timeline and slides on the models proposed by various key figures in the atomic theory timeline. How a scientific explanation is conditional but may become more convincing when predictions based on the model or theory are confirmed later on by further ideas/scientific data. The PowerPoint covers how a theory may change as new evidence is found. He imagined the equation of evolution of the wave function associated with the state of a particle, he allowed the development of the theoretical formalism of quantum mechanics.This extra lesson is not explicitly covered by the Cambridge iGCSE syllabus but it makes a nice addition to the course, as project work, an introduction to the idea of scientific models or just as an introduction to the periodic table or atomic theory.Įach PowerPoint contains a lot of detail, thus this lesson is not really intended to be delivered in the presentation format as like my other lessons. The alpha particles passed through the gaps but bounced back from the dense, positively charged nucleus.Īustrian physicist Erwin Schrödinger is one of the founders of quantum mechanics, but he’s most famous for something he never actually did: a thought experiment involving a cat.

Rutherford concluded that atoms consisted largely of empty space with just a few electrons, while most of the mass was concentrated in the centre, which he termed the nucleus. But while most of the particles did pass through, some bounced right back, suggesting that the foil was more like a thick net with a very large mesh. The effect would have been like a bunch of tennis balls punching through a thin paper screen. Under Thomson’s model, the atom’s thinly dispersed positive charge would not be enough to deflect the particles in any one place. He decided to investigate atoms more closely by shooting small, positively charged alpha particles at a sheet of gold foil. Rutherford is known as the father of the nuclear age. Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 4 ), is a fundamental concept that states that all elements are composed of atoms. He added that electrons move simultaneously behaved like waves, not being confined to a particular point in space. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 1.5.4 1.5. He affirmed that electrons orbit the nucleus at fixed energies and distances, able to jump from one level to another, but not to exist in the space between. In 1913, Niels Bohr expanded on Rutherford’s nuclear model. This discovery end in nuclear fission and, therefore, the atomic bomb… He received a Nobel Prize in 1932.īritain physicist who has discovered the neutron in 1932 and received thereafter a Nobel prize in 1935. He showed with the “uncertainly principle” that it was impossible to determine both the exact position and speed of electrons as they moved around an atom. Lorem ipsum dolor sit amet, consectetuer adipiscing elit, sed diam nonummy nibh euismod tincidunt ut laoreet dolore magna aliquam erat volutpat. Then, he received many honors for his work. He concluded that the various compounds were combinations of atoms of different elements, each of a particular size and mass, that could neither be created nor destroyed. Dalton showed that common substances always broke down into the same elements in the same proportions. Then, elements combine to form chemical compounds and that in chemical reactions atoms are combined separated or rearranged. He published, in 1808, five atomic laws which are: -All matter is composed of extremely small spherical indivisible particles called atoms. Thomson won a Nobel Prize in 1906 for his electron discovery, but his model of the atom didn’t stick around long.Įnglish chemist and physicist. In what we might call the chocolate chip cookie model of the atom, he showed atoms as uniformly packed spheres of positive matter filled with negatively charged electrons. Physicist who in 1897 discovered the electron.

Its name has been given to a unit to express the amount of electrical charge: the Coulomb. He later stated Coulomb's Law, which governs the interaction of electrical charges. In 1757, he invented the torsion balance, which allowed the force of magnetic and electrical attraction to be measured. He discovered that two opposing charges attract each other. In 1789, Coulomb continued his research on magnetism, friction and electricity. His ideas were opposed by the most popular philosophers of his day who stating instead that matter was made of four elements: earth, wind, water and fire. He called these particles “Atomos”, Greek for indivisible. And he even speculated that they vary in size and shape depending on the substance they compose. Democritus first proposed that everything in the world was made up of tiny particles surrounded by empty space. Democritus is one of the preceptors of materialistic theory.
